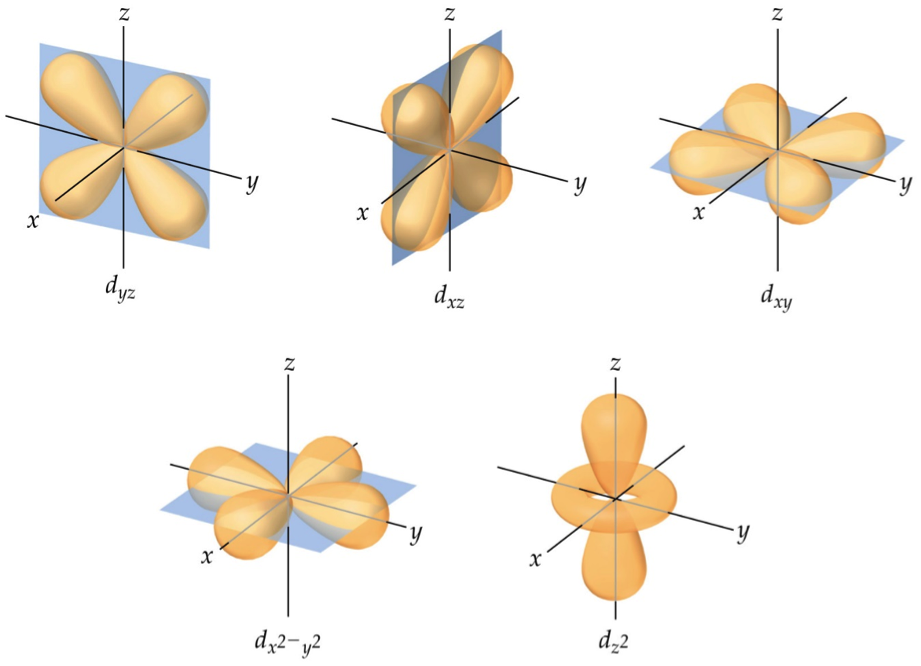 
We include specific suggestions for how VSEPR-style rationalizations of molecular structure can be replaced with more accurate conceptions of hybridization and its relationship to electronegativity and molecular geometry, in accordance both with Bent's rule and the consistent features of modern wavefunctions as exhibited by natural bond orbital (NBO) analysis. The angle between any two bonding pairs in the tetrahedral electron-group arrangement is 109.5. VSEPR-style conceptions thereby detract from more accurate introductory-level teaching of orbital hybridization and bonding principles, while also requiring wasteful ''unlearning'' as the student progresses to higher levels. with four BP, has a tetrahedral molecular shape. VSEPR-style representations of orbital shape and size are shown to be fundamentally inconsistent with numerous lines of experimental and theoretical evidence, including quantum mechanical ''symmetry'' principles that are sometimes invoked in their defense. For example, based on the shapes and orientations of the 2s and 2p orbitals. orbitals are mixed and rearranged to give a new set of four. The VSEPR model, is a simple model used for predicting molecular shape. Both C atoms have one 2p orbital that has yet to be used for bonding.We describe the logical flaws, experimental contradictions, and unfortunate educational repercussions of common student misconceptions regarding the shapes and properties of lone pairs, inspired by overemphasis on ''valence shell electron pair repulsion'' (VSEPR) rationalizations in current freshmanlevel chemistry textbooks. The main postulates of VSEPR theory are as follows: The shape of a molecule depends upon the number of valence shell electron pairs (bonded or nonbonded) around the central atom. 52 1.9K views 2 years ago Organic Chemistry Lessons VSEPR Theory helps us understand molecular geometry, where orbital hybridization explains how our atomic orbitals create the geometry to. Unit 2 2.7 VSEPR and Bond Hybridization 5 min read decemA Anika P D Dalia Savy AP Chemistry 269 resources See Units Valence Shell Electron Pair Repulsion (VSEPR) Lewis structures can determine properties such as geometry, bond orders, bond lengths, and dipoles for molecules. To be able to use VSEPR theory to predict shapes, the molecule first needs to be drawn in. 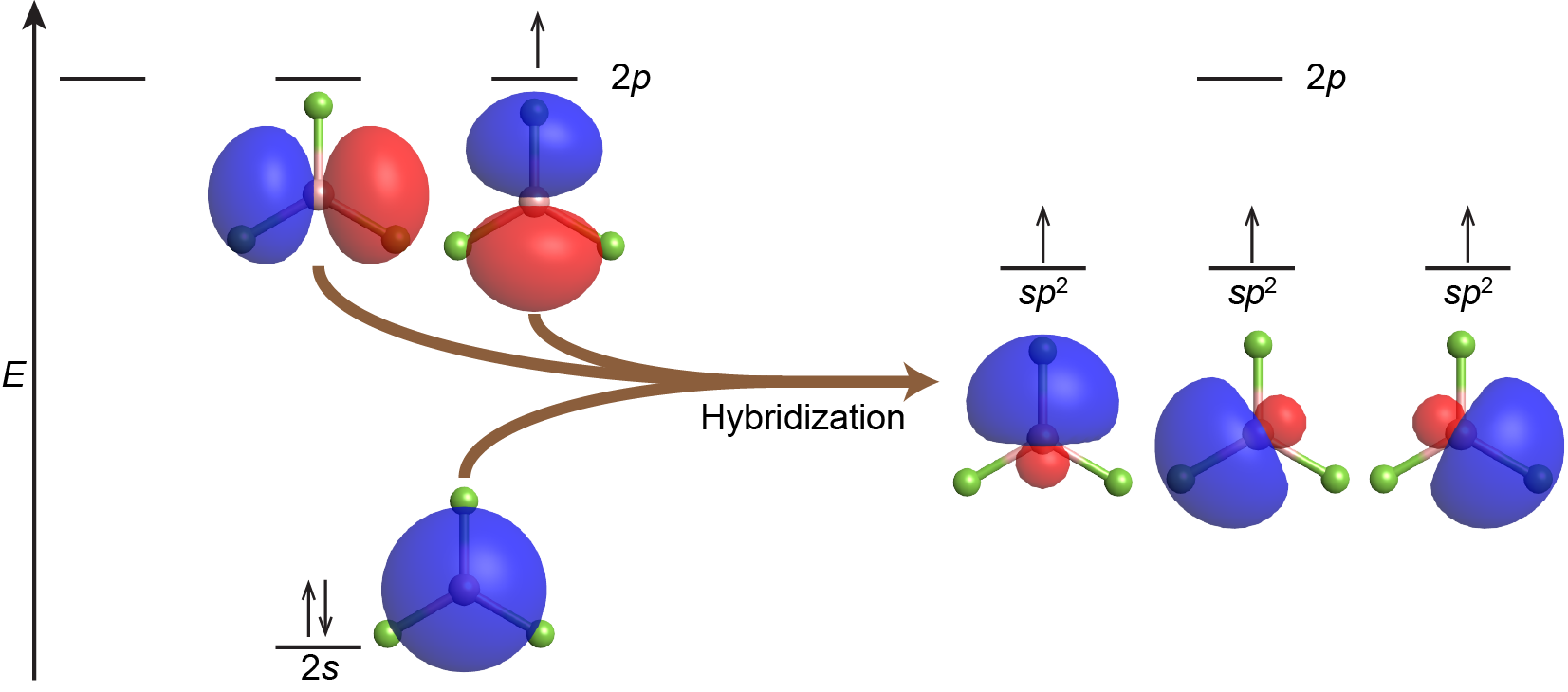
The third hybrid orbital on each C atom can overlap to form a sigma bond between the two C atoms. electron domain geometry linear A A generic atom - angle between e - domains is possible molecular geometries a) Linear - only linear geometry is possible with two electron domains Example: Bel2 l Be l Example: 2 - Note: nly two electron domains around central atom since multiple bonds count as a single domain. The VSEPR theory describes five main shapes of simple molecules: linear, trigonal planar, tetrahedral, trigonal bipyramidal, and octahedral. 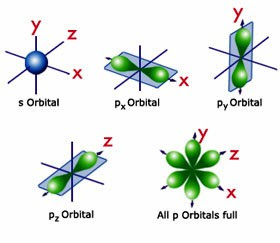
Two of the sp 2 hybrid atomic orbitals on each C atom can can overlap with the s orbitals on two H atoms. Determine the shapes of molecules using hybridized orbitals, linear combination of atomic orbitalsmolecular orbitals (LCAO-MO), and valence shell electron. Combining one ns and two np atomic orbitals gives three equivalent sp 2 hybrid orbitals in a trigonal planar arrangement that is, oriented at 120° to one another.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
